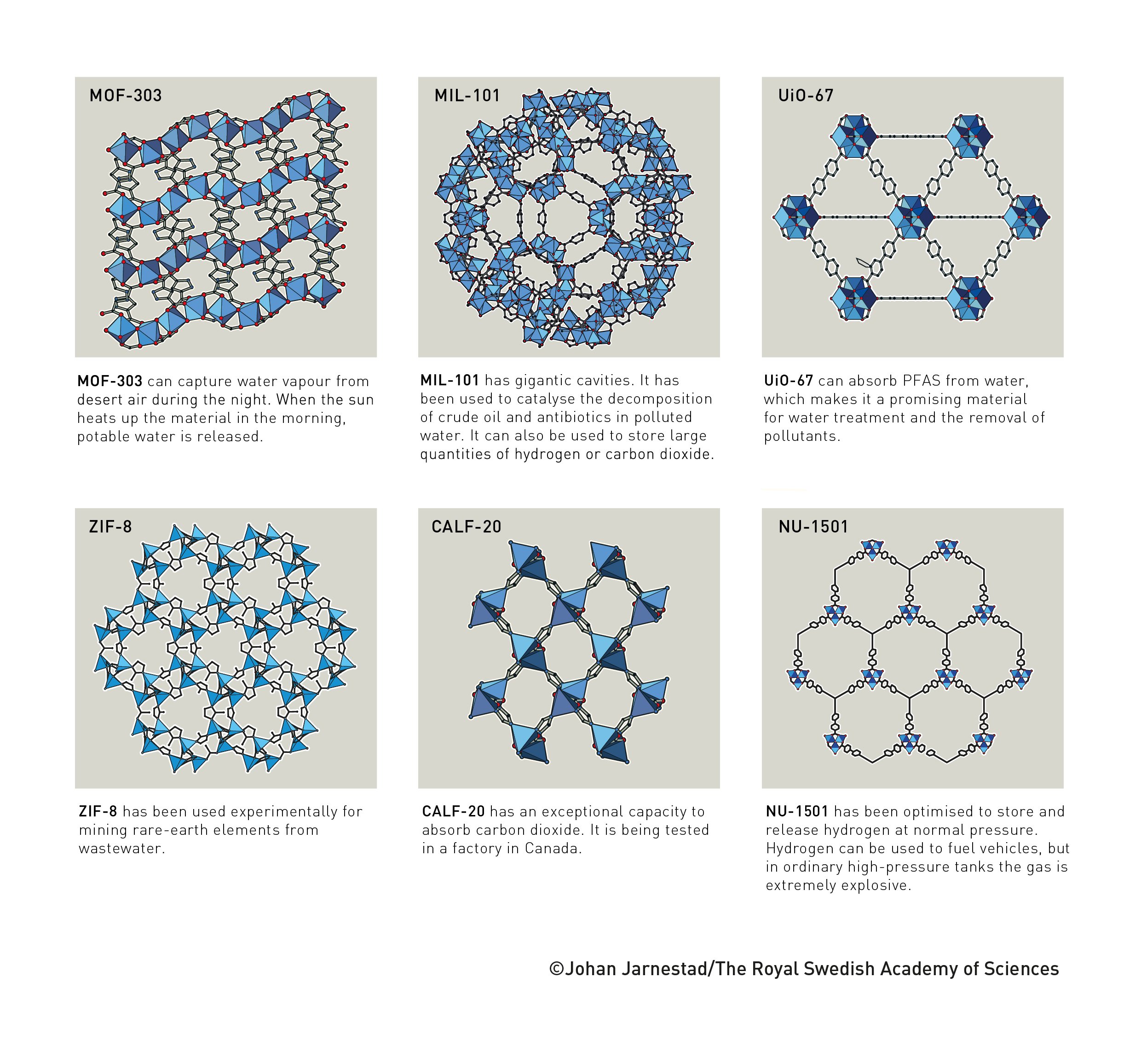
The polymers around us are basically a disordered mess, with long chains of atoms tangled around each other. But starting around 1990, chemists began developing techniques that allow us to build polymers with precisely defined structures. These polymers, called metal-organic frameworks (MOFs), have distinct chemical properties: large pores that can be used to filter or store gases, catalytic centers within the polymer, and more.
On Wednesday, the Nobel Prize Committee honored three researchers for their role in developing MOFs: Richard Robson for demonstrating the first MOF, and Susumu Kitagawa and Omar Yaghi for developing them to their full potential.
Building a structure
Most polymers are built from individual molecules that are linked together by flexible bonds, allowing the molecules to flop around. As a result, in their final form as something like a plastic bag or a bike tire, the polymers are a complicated tangle, with molecules wrapped around each other in no particular order. We can still control some aspects of the resulting polymer by manipulating its bulk properties or changing the molecules it’s built from, but there’s not a lot of chemistry we can do beyond that.
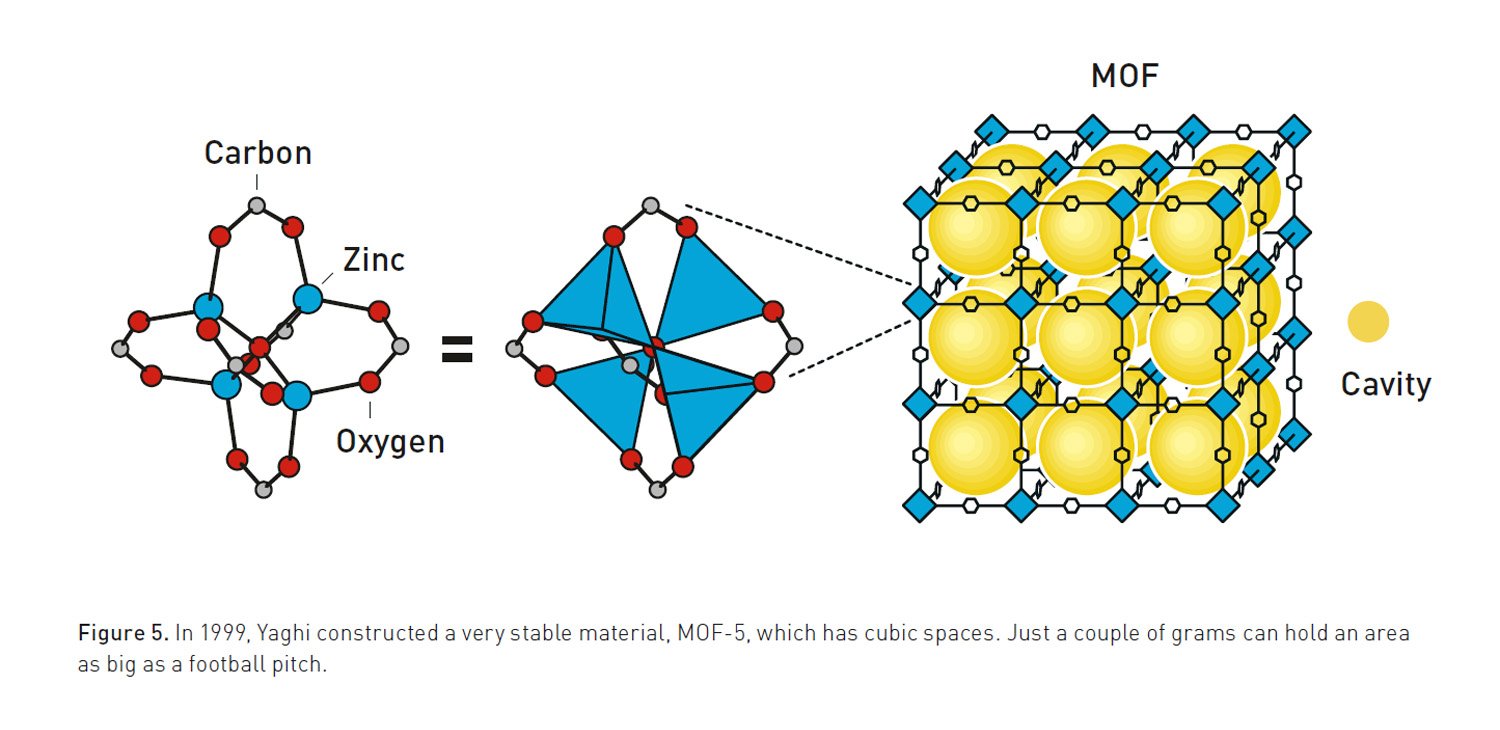
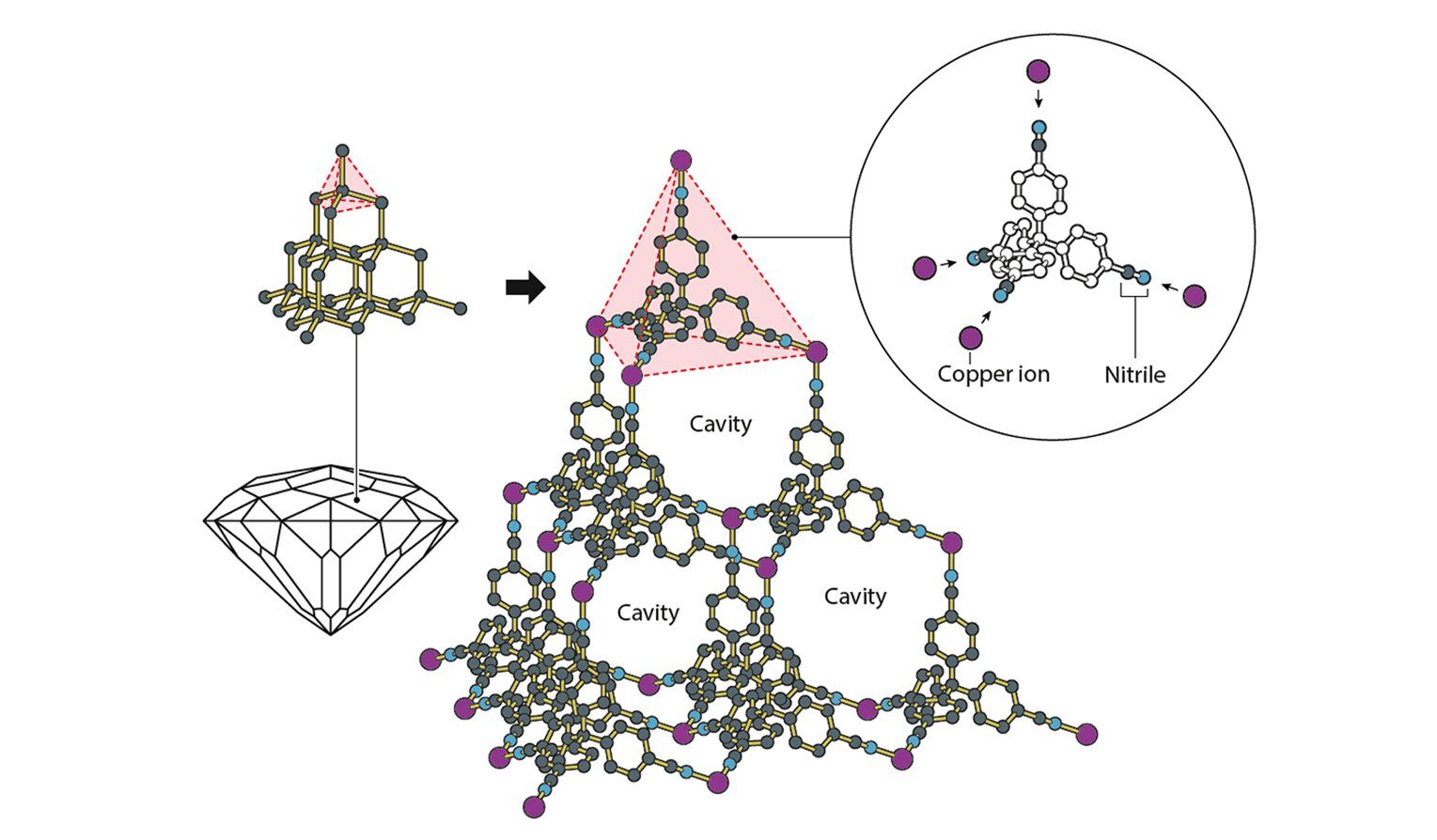
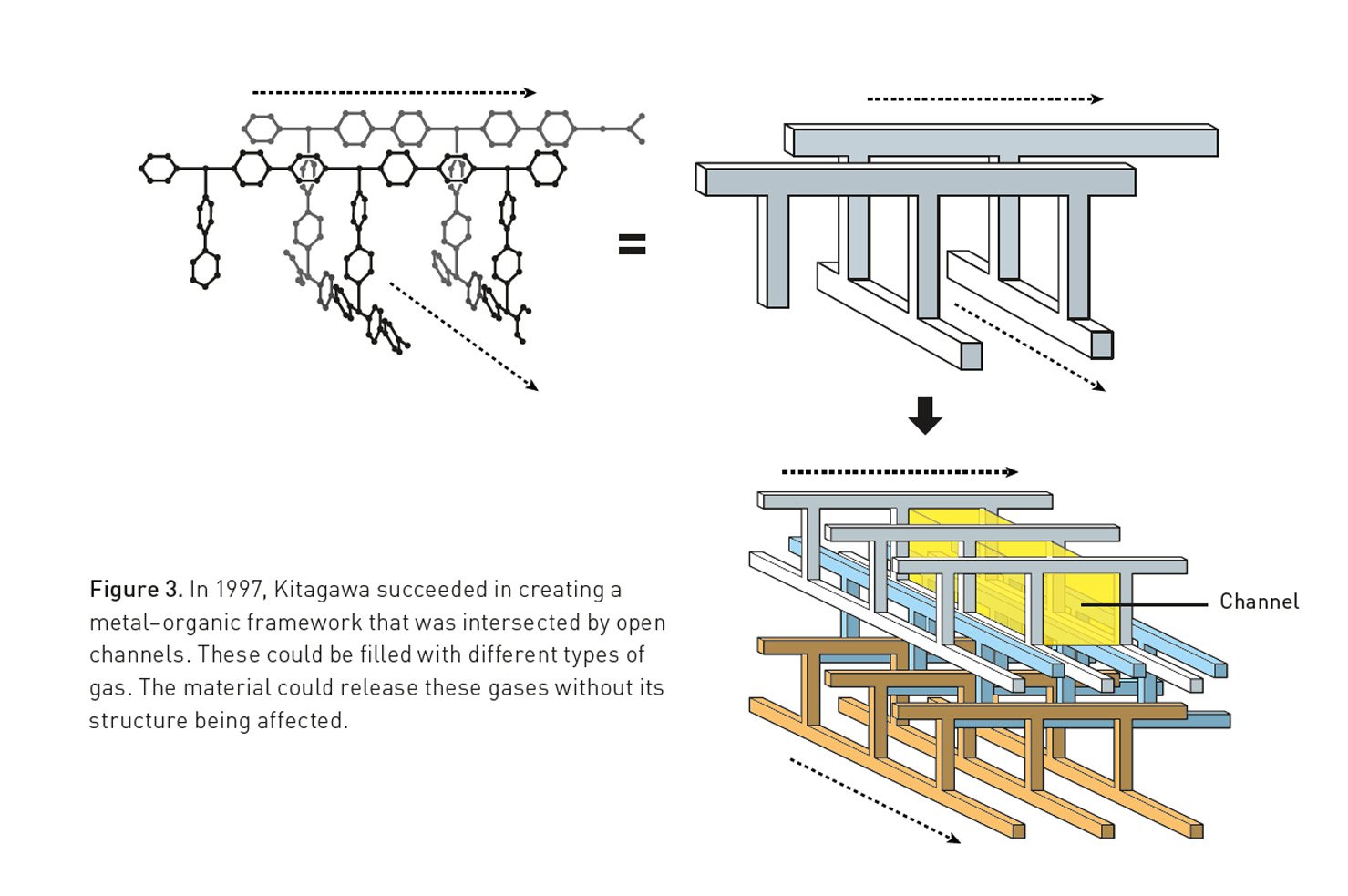
With MOFs, the metal portion of things allows us to control the geometry of how molecules get linked together. The metals act a bit like hubs, with rigid organic molecules extending out in multiple directions. Because of the nature of their orbitals, metals can bond with a specific number of organic molecules, and each of those bonds has set angles between it and all the other bonds. So by choosing a metal carefully, it’s possible to craft specific three-dimensional structures, with metals and organic molecules alternating with each other.

 Loading comments...
Loading comments...
